SHAM CAM REGULATORY WORKSHOP WITH MEDICINES CONTROL COUNCIL
SHAM CAM REGULATORY WORKSHOP WITH MEDICINES CONTROL COUNCIL
MCC throws cams to pharmaceutical industry under opaque international harmonisation scheme
Anthony Rees and Sue Schonken
(Executive Committee members of the TNHA)
29 June 2016
On Monday the 13th of June the TNHA attended a ‘Regulatory Workshop’ in Midrand, co-hosted by the Health Products Association (HPA) and the Self Medication Association of South Africa (SMASA).
The TNHA attended as observers and did not participate in the workshop, knowing that our attendance and engagement would have been construed as our tacit acceptance of the current regulatory system for ‘complementary medicines’ which we reject.
At the workshop officials from Medicines Control Council (MCC) and Department of Health (DoH) were invited to outline and discuss the long-overdue regulatory amendments for complementary medicines, and provide details about the new category of complementary medicines referred to as ‘health supplements’.
What ensued was highly problematic.
NO REGULATIONS RELEASED BEFORE WORKSHOP
When the workshop was first announced more than two months ago, the MCC indicated that revised complementary medicine regulations would be gazetted for public comment, and that accompanying guidelines would be released well in advance of the workshop. This would have afforded stakeholders adequate time to study their contents and ask pertinent questions.
On the last working day (Friday) before the workshop people in the industry were desperately looking for copies of the regulations and guidelines, having not yet received copies. The TNHA urgently made enquiries with Parliament and the Government Printer without any success. Nothing had been gazetted.
Failing this, one would have expected these documents to be distributed on the day of the meeting, but they weren’t. The MCC’s failure to provide working documents effectively crippled any effort by the attendees to present in time, their list of questions, which the MCC had stipulated as a pre-condition of the workshop.
On arriving at the workshop empty handed, delegates were informed that four new “guideline documents” [Doc 1] [Doc 2][Doc 3] [Doc 4] had been released on the MCC website at 1:20 am on the morning of the workshop, and that the anticipated regulations were “delayed” and would still to be gazetted ‘imminently”. We ask: What government department is open for business and publishes official documents on its website an hour after midnight over a weekend? What was the agenda behind this?
In her opening address to the workshop Ms Joey Gouws (MCC Registrar of Medicines) stated that the regulations were delayed because the 24-member Medicines Control Council had met only two days prior to the workshop, and had not had sufficient time to approve and release them. She claimed they would be released imminently: More than two weeks has passed since the workshop (at the writing of this report) and still no regulations have been gazetted.
For the past nine months the standard response of Gouws’s office to any queries involving the expected date of release for the amended regulations has been met with the ‘imminently’ reply. We have heard this ‘imminently’ word for months on end and quite honestly question if the MCC understands the Oxford Dictionary meaning of imminent ‘very soon’.
One would have reasonably anticipated, as attendees of this technical workshop that the MCC, in full knowledge of their failure to publish in good time for the workshop, would have made workshop packages ready at the entrance so that all parties had appropriate material to review. This did not happen.
The delegates sat with no material whatsoever which they could refer to and make notes on.
RECORDING OF PRESENTATIONS DENIED
When we enquired with the Workshop Coordinator if the presentations were being officially recorded on video or audio, we were informed that the MCC had expressly requested that no recording of the workshop be allowed, including by delegates.
If the MCC embraced transparency or valued stakeholder participation, they would not have imposed this draconian prohibition.
QUESTIONS FROM THE FLOOR DENIED
We were informed prior to the workshop that questions would not be allowed to be asked from the floor and that questions directed to the MCC/DoH Panel had to be submitted in writing and sent to the workshop coordinator at least one week in advance.
In her welcome speech, workshop moderator Dr Konji Sebati reiterated this rule, stating that delegates would be flatly ignored if they interjected during presentations, or attempted to pose any questions.
Sebati is the CEO of the Innovative Pharmaceutical Association of South Africa (IPASA) and Director of the Department of Traditional Knowledge and Global Challenges at the World Intellectual Property Organization (WIPO). She previously represented South Africa as ambassador to Switzerland, France, The Vatican and Liechtenstein. She also worked for Roche Pharmaceuticals and served as Medical Director of Pfizer.
IPASA represents Western drug companies conducting business in disease management in South Africa, and represents 43% of the local pharmaceutical drug market. In 2014 IPASA was described by Health Minister Aaron Motsaledi as being behind “Big Pharma’s satanic plot of genocide” in South Africa, following a leaked document, allegedly drafted by IPASA describing how “astroturf (false grass roots) organizations” would be created by Big Pharma to exert pressure on the SA government to change its policies directed at eliminating and bypassing intellectual property rights (patents) for costly pharmaceutical drugs.
Just how a complementary medicine regulatory workshop could be refereed by the CEO of Big Pharma’s official representative in South Africa, is beyond questionable, and telling of what forces may be pulling the MCC’s strings in what has become the pharmaceutical expropriation of natural health products.
WRITTEN QUESTIONS CENSORED
At the end of the lunch break, after most delegates had returned to the lecture theatre we witnessed Ms Gouws and Dr Sebati screening the question sheet submitted by attendees a week prior to the meeting, and discussing which questions would be dealt with by the speaker panel at the end of the programme. Gouws crossed out questions related to issues they she and her colleagues were no doubt not prepared to answer.
This was blatant censorship of valid questions raised by concerned delegates.
DRUG REGULATORY HARMONISATION SCHEME BEHIND CAM REGULATIONS
In her opening lecture, Ms Gouws pointed out that the recent regulations (which will severely restrict the public’s access to natural health products and traditional medicines in South Africa) are in alignment with the MCCs regulatory convergence (harmonization) programme under the auspices of the International Coalition of Medicines Regulatory Authorities (ICMRA).
View Slide 21 of Gouws’ presentation as evidence.
Over the past two years we have been researching ICMRA, run by a few dozen unelected bureaucrats, who meet on average twice a year in various foreign locations, making decisions for the health of billions of people on seven continents behind closed doors with no transparency or consultation.
Who are these people? Their names aren’t provided in their press releases or on their website. Who pays them? What authority do they have?

Photo: Meeting in Amsterdam, Netherlands (3-6 December 2013) where global drug regulators agreed to the establishment of ICMR.
On her return from an ICMRA meeting held in London in early 2014, Dr Gouws reported to local industry insiders that ICMRA Member Countries had agreed that various countries, including South Africa would follow Health Canada’s lead in bringing natural health products under a quasi-drug-regulatory model, and that these regulatory schemes would become benchmarks for the other countries to follow. South Africa and New Zealand appear to be the first dominos to fall in this global strategy.
We would like to know why our legislature (Parliament) has seemingly never been briefed about or approved this scheme.
WHAT IS ICMRA?
The conceptualisation of ICMRA was the brainchild of the previous US Food and Drug Administration Commissioner, Margaret Hamburg. (see “New Realities of Globalization — Implications for Health, Medicine and the Role of the Regulator“, presented in London on the 6 March 2014. [Ref 1][Ref 2]
ICMRA describes itself as an executive-level and strategic co-ordinating entity of regulatory authorities, which aims to provide the global architecture for pharmaceutical drug regulation. It also aims to promote the equivalence and convergence (harmonization) of national and regional regulatory systems.
ICMRA does not have a permanent office. Its Secretariat functions “virtually” on an interim basis and is currently chaired by Health Canada’s Health Products and Food Branch (HPFB-HC).
This HPFB-HC is responsible for natural health product regulations, and currently influencing the natural health product regulatory schemes in South Africa and New Zealand.
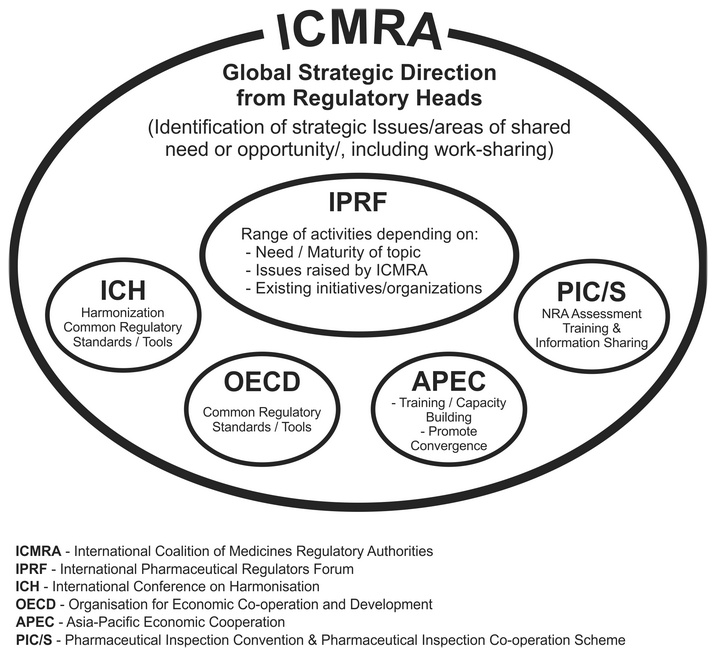
ICMRA’s membership is voluntary, and currently comprises the drug regulatory authorities from the largest pharmaceutical export and import nations, including Australia (TGA), Brazil (ANVISA), Canada (HPFB-HC), China (CFDA), Europe (EMA and DG-SANCO), France (ANSM), Germany (PEI), Ireland (HPRA), Italy (AIFA), Japan (PMDA and MHLW), Mexico (COFEPRIS), Netherlands (MEB), New Zealand (Medsafe), Nigeria (NAFDAC), Singapore (HSA), South Africa (MCC), South Korea (MFDS), Switzerland (Swissmedic), United Kingdom (MHRA) and the United States (FDA). The World Health Organization (WHO) is the only participating observer organization.
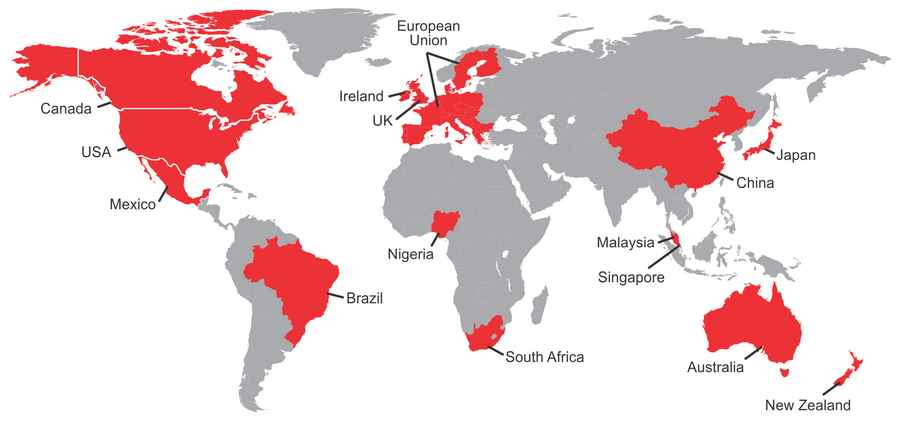
Map of current ICMRA Member Countries (June 2016)
The ICMRA is currently in its interim phase (2014 – 2016) and is expected to expand its membership to more countries beyond 2017. With footholds in influential threshold countries on every continent, ICMRA harmonized countries will become benchmarks for other neighbouring countries and regional regulatory agencies to follow, for example the soon-to-be lunched African Medicines Agency (AMA).
Natural health freedom activist groups like the TNHA around the world will need to monitor this shadowy decision-making body more closely in the future, and demand more transparency at its meetings. ICMRA must open itself up to participation by key stakeholders and civil society organisations, or it must be dismantled for having an illegitimate mandate.
While the world’s attention has been fixed on the Codex Alimentarius Commission over the last two decades, and the potential threat it poses by limiting access to nutrients and functional foods, ICMRA has been quietly established with greater potential to undermine our health freedoms in a very short time frame, and with no accountability.
QUESTION: Who is actually making decisions for natural products in the world? Pharmaceutical drug regulators? Are we being bulldozed by alliances and agreements we know nothing about?
MUSINGS ON THE “WORKSHOP”
The Oxford Dictionary defines a workshop as: “A meeting at which a group of people engage in intensive discussion and activity on a particular subject or project”. And “Present a performance (concept) of a … work, using intensive group discussion and improvisation in order to explore aspects of the production prior to formal staging.”
In essence we believe that any workshop has as its purpose to review the subject matter, break into intensive discussion groups, explore meanings and implications, make input towards qualified understandings and interpretations, and then present the input of the work groups into the larger group for serious consideration by the workgroup presenters and officials.
Instead, what the delegates at this workshop were subjected to was sitting in restrained silence like Grade 2 children, under threat of personal embarrassment or eviction, being presented with a collection of out-of-date, condescending lectures by bureaucrats and pious orthodox medicine academics (all slanted towards benefitting pharmacists and chemical drug companies) who were all of the conviction that ‘a medicine is a medicine is a medicine’ and that there was no difference between orthodox pharmaceutical chemical drugs and natural health products. From their lecterns they decreed that there is little difference in the way natural health products and pharmaceutical drugs should be manufactured and assessed for registration.
Clearly, there is a critical deficiency in knowledge among MCC officials to the fundamental tenets of traditional and natural health products, and the paradigm to which they belong, let alone the inherent safety of these time-tested health regimens.
This elective blindness is married to a blunt refusal to grasp the merits of these products. What a joke (if it wasn’t so serious), a farce, a charade. This was an insult to the collective intelligence of the natural health product industry delegates present.
In contrast, the pharmacists attending from the pharma-drug industry, or who work as private registration consultants, appeared to be pleased with the presentations. No surprises here, as they are already jammed into the sausage-mill of over-regulation, and rubbing their hands at the financial opportunities that regulation of complementary medicines sector lands of their laps.
The sub-text here is that the beneficiaries of these proposed CAM regulations would be pharmacists, who will have to be permanently employed by manufacturers, as well as private medicines registration consultants.
The only “concession” in all the tirades of irrelevant and biased deliveries of six hours of sound-bytes by the presenters, was an envisaged separation of natural health products into “Low Risk” and “High Risk” products and dossiers, which related only to the “health claims” or inferred usefulness of the natural products, and had nothing to do with any safety issues of the products.
The very notion of “claims” on products is so broad and so obtuse, that even if somewhere in the world there existed some reference to a natural health substance’s usefulness in preventing or treating any single disease condition, any products (or applications) containing these compounds, would default to the “High Risk” category, meaning their scheduling and removal from shelves.
There was no suggestion that genuine professionals in the natural health products industry would be consulted, as all MCC dossier assessments would be placed before a panel of pharmacists to arbitrarily determine High vs Low Risk categorisation, or whether a dossier can proceed to the next step of the registration process.
What is evident is that a small homeopathic clique on the Complementary Medicines Committee (CMC), a single profession represented by the Allied Health Professions Council, appears to currently have a marginal degree of favour within the MCC, offering thinly veiled “lip service” to the CAMS sector.
This is not legitimate representation of the broad-based traditional and natural health sector of the Nation.
How is it that a few people can make unilateral decisions for a whole sector which includes doctors who practice with integrated health strategies, nutritional health practitioners, the manufacturing industry and millions of health conscious consumers?
THE INEXPLICABLE INFLUENCE OF THE ALLIED HEALTH PROFESSIONS COUNCIL
One of the MCC members who delivered a lecture, Prof Andy Gray reiterated that all herbal and homeopathic products sold in South Africa are defined as ‘discipline-specific’ complementary medicines, and would not be included in the new ‘food supplement’ category, rendering them difficult to register for over-the-counter sale.
The rationale behind this decision is that these substances and products are attributed to the medicines described in the exclusive scope of practice of Allied Health Professionals registered with the Allied Health Professions Council, in terms of the Allied Health Professions Act (Act No 63 of 1982).
This is absurd and irrational, as the majority of commercial herbal and homeopathic products have never been supplied or manufactured exclusively by these practitioners. The majority of these products (by market share) have been historically sold in pharmacies, health stores and by direct marketing companies.
The percentage of sales for homeopathic and herbal products to practitioners registered with the Allied Health Professions Council makes up a small percentage of gross sales of natural health product manufacturing companies. Practitioners registered with the Allied Health Professions Council are NOT the innovators of these products, and have no inherent right to exclusivity of them, considering most are demonstrably low-risk substances.
Allied Health Professionals are not, according to the Allied Health Professions Act, allowed to own companies trading in medicines they prescribe.
The Allied Health Council is on record as being happy with the current MCC regulatory scheme, and in fact its leadership have been co-opted exclusively to the MCC’s Complementary Medicines Committee (CMC). One homeopath, Dr Neil Gower has even been appointed to the 24-member Medicines Control Council and has been a key architect in drafting the regulations.
Under the current regulatory scheme the Allied Health Professions Council will monopolise the CAM sector. It supports the removal of effective, herbal and homeopathic products from the freely available (over-the-counter) market, wishing to dispense and sell them exclusively from their dispensaries, after paid consultations.
Because most complementary medicines (‘Category D’ products) are now hinged to the disciplines represented by the Allied Health Professions, the sustainability of such categories are dependable on the sustainability of the Allied Health Professions currently represented.
At present the Allied Health Professions Council has a major problem: It has very few students graduating in the professions of Phytotherapy (Medical Herbalism), Naturopathy, Traditional Chinese Medicine, Unani Tibb and Naturopathy who are eligible to enter onto the Council’s professional registers. The only university in South Africa that provides training in these professions, namely the University of the Western Cape, has experienced an ever-dwindling number of fourth and fifth-year graduates.
A few years ago, the professional register for Ayurveda (Indian traditional medicine) was closed due to the closure of the only training programme at UWC, and no graduates being admitted to the profession register. Yet, for some inexplicable reason, Ayurvedic health products are sub-categorised as ‘discipline specific’ complementary medicines.
This year the TNHA has ascertained that no more than eight students will enter their fourth year of training across the four professions represented. We are aware that many of the previous graduates of the UWC programme no longer practice or have never opened practices. Most graduates work as reps for medicines manufacturers, labour in health shops or have changed careers.
The net result of the failure of these training programmes will inevitably result in the non-sustainability of the UWC courses in the near future, and the potential closure of the registers for these professions, a repeat of what happened in 1986.
Many of the existing practitioners currently on the Allied Health Professions Council’s registers were registered between 1999 and 2002, after the registers were re-opened by Health Minister Nkosazana Zuma after two decades of being closed. Those admitted to the registers at that time were predominantly practitioners in the 40 to 50 year-old age group, and who are expected to retire in the next few years. With the exception of Homeopathy, the future of the prescribing professions of the Allied Health Professions Council looks dire, and as a consequence of this, so may the ‘discipline specific’ sub-categories of complementary medicine described in the MCC regulations attached thereto.
If the professions are no longer practiced or represented, what will become of the MCC ‘discipline specific’ categories legally tied to them?
MCC CLAIM: A MEDICINE IS A MEDICINE IS A MEDICINE
The one lecturer openly declared this to be a “fact”, with a bravado that was intended to subvert any possible challenge to his interpretation. What of this claim? Reality check: A DRUG IS A DRUG IS A DRUG, AND MANY CARRY SERIOUS POTENTIAL AND/OR REAL HARM.
This interpretation that “a drug is a drug is drug” has generally been voiced in the corridors of the MCC and the DoH to justify their inappropriate and harsh stance towards natural and inherently safe natural health products.
INAPPROPRIATE CTD DOCUMENTATION FOR REGISTRATION
The MCC is still demanding that all applications for complementary medicines and food supplements be supported by a completed Common Technical Document (CTD), colloquially known as the ZACTD. This document (or dossier) is required to be completed by a registered pharmacist, responsible for the manufacture of complementary medicines and food supplements.
The document is highly technical in nature and may take hundreds of costly hours to complete and obtain sufficient documentary clinical trial evidence in support of a single product application.
We believe the ZACTD is appropriate for high risk pharmaceutical drugs, but not for natural health ingredients or mixtures thereof. For a single ingredient application, the ZACTD may exceed 250 pages of highly technical details and jargon, and becomes exponentially more voluminous the more ingredients are included in a product.
Here is an example copy of a completed CTD dossier mentioned by workshop presenter Estelle Taute, Director of Operations and Administration at the MCC. Why is this necessary?
Having to provide all this information for multi-ingredient products and/or an entire range of products requiring registration is ludicrous.
GOOD MANUFACTURING PRACTICE (GMP) OVERKILL
One of the ten DoH Inspectors of Law Enforcement Unit, Andrea Julsing Keyter, gave an hour long presentation on the GMP requirements for manufacturing natural health products. The first twenty minutes could have been interpreted as a sales pitch for multi-million rand air-conditioning and laminar airflow management systems which are required to be installed in all MCC licensed manufacturing facilities. This week, we contacted the owners of two small natural health product companies who recently obtained quotes for these compulsory ‘HVac systems’. The cheapest installation came in at R12-million.
The MCC also contends that all manufacturers who currently manufacture natural health products should already be licensed, or have applications lodged. They expect Site Master Files to be completed and critical processes validated to ensure consistency and compliance with specifications.
They require ‘Good Documentation Practices’, where all raw ingredient handling, manufacturing and post-processing instructions and procedures are recorded and that qualified operators (pharmacists) in the production facility are trained in document procedures. A copy of the WHO guidelines for pharmaceutical GMP which the MCC adheres to can accessed from the link below
http://www.who.int/medicines/areas/quality_safety/quality_assurance/TRS986annex2.pdf
CAMS INDUSTRY IS NOT FALLING FOR THE MCC SUBVERSION END-GAME
Essentially, there was nothing new to hear and learn at this workshop. Even the disdain and disregard for participants are old hat.
The regulations are not implementable.
Dr Gouws admitted in her presentation that over 120 000 natural health products had been audited by the MCC between 2002 and 2011. Of these, only 110 (that’s right – one hundred and ten), or 0,09%, have been submitted in terms of the 2013 call-ups for evaluation and possible processing to towards registration. In the end, of the 28 final submissions selected in the first sifting, only six have passed the next screening process allowing for them to be evaluated for their safety quality and efficacy. Of these six products, three are manufactured by offshore companies, and three are locally produced. We not even sure if these six products have a chance of reaching the end-phase of registration.
These carefully orchestrated MCC regulatory actions thus far are tantamount to the obstructive removal of the vast majority of health products not manufactured by drug companies from the market.
It’s clear that those products which will be allowed to remain on health store shelves or through direct selling systems after 2019 will be low-potency nutrient supplements and products with no curative or preventative health claims. Like in Canada, the post-regulatory marketplace will be littered with cheap, mostly synthetic multivitamin type products and single substances (herbs and homeopathics) with no indication of health benefits being allowed to be expressed. In the vacuum created by the loss of high potency, innovative and integrative products (the products that work), drug companies and large chain store pharmacy companies will take advantage and offer their junk products as the only choice to consumers.
Although there is an expectation among Allied Health Practitioners that the high potency, higher scheduled and integrative products will shift into their dispensaries in terms of their prescribing rights, companies will no longer manufacture or import these products due the economies of scale (catering for a small sector), and subsequently having no means of cost recovery after having to comply with the onerous and costly compliance and licencing costs to stay in business.
In the end, the manufacturers, wholesalers, retailers, direct marketers, prescribing practitioners and the public will lose. The only winners will be the pharmaceutical industry.
#REGULATIONS MUST FALL
The natural health sector represented by the TNHA is resisting this unconstitutional and ultra vires attempt by the MCC to subvert the enormous value of the health products sector in our country.
Dr Gouws declared the MCC capacity for evaluating all medicines (pharmaceutical and CAMS products) to be 320 products per year. In 2009 a Ministerial Report highlighted that over 9 500 pharmaceutical drugs which are being prescribed or sold over-the-counter in South Africa have not been trialled or appropriately tested for their safety or efficacy. These are potentially high-risk products. The most generous computation for this failure of capacity in the MCC or DoH, implies that the MCC needs 200 years to for registering CAM products. In other words … ‘impossible’.
The TNHA rejects the entire regulatory system as per the 2013 regulations for natural health products on the basis that they are:
- ULTRA-VIRES (have no legal standing). The MCC does not have a clear legislative mandate to expand its scope beyond pharmaceutical medicines. In other countries where these products have been regulated, the legislators drafted and passed the laws for these low risk products. The Medicines and Related substances Act is deficient in enabling provisions for this these new systems of medicine. If different health professions (orthodox, allied and traditional) have separate legislation and statutory councils to represent their professions and philosophies, so must their medicines.
- DISCRIMINATORY. The regulations for complementary medicines do not include African Traditional Medicines and therefore are in breach of Section 9 (Equality clause) of the Bill of Rights in the Constitution. Herbal medicine is herbal medicine. There cannot be regulations enforced for non-indigenous traditional disciplines (Traditional Chinese Medicines, Traditional Western Herbal Medicines, Traditional Unani Tibb Medicines and Traditional Ayurvedic Medicines) and no equal regulation for indigenous African Traditional Medicine, used by the majority of South Africans. The discrimination is unfair, until proven to be fair.
- UNDERMINING OF NATIONAL SOVEREIGNTY. Implementing unconstitutional harmonisation agreements (ICMRA/WHO etc) undermines our local law-making process and national sovereignty.
- BREACH OF THE ADMINISTRATIVE JUSTICE ACT. Serious breaches of due law-making process have occurred since 2008. There has been no transparency or meaningful consultation in the manner regulations have been drafted and gazetted.
- ANTI-COMPETITIVE. The CAMS regulations create an anti-competitive environment favouring the pharmaceutical sector, necessitating a Competition Commission investigation.
- ILL-CONCEIVED. The regulations are irrationally pegged to the disciplines represented by the Allied Health Professions Council, with no international precedent or rationale for this interplay. Allied health practitioners are not the innovators, manufactures or sellers of these products to the public, nor do they have a vested right to their exclusive distribution.
- DISPROPORTIONATE. The MCC was established in 1965 to control and regulate high-risk pharmaceutical drugs in South Africa and to protect the public from harm. It, and other drug regulators around the world have failed to achieve this mandated instruction by their respective legislatures. This is evidenced by the fact that pharmaceutical drugs are now the third leading cause of death in the industrialised word. This is followed by thousands of deaths caused by common foodstuffs and common household chemicals. Commercial natural health products are extremely low risk products, with no verifiable, recorded deaths in South Africa from their use in the 50 years the MCC has been in existence. The disproportionate regulatory fervour and resources expended on CAM regulation verses high risk drugs is irrational.
- NON-REPRESENTATIVE OF THE CAM SECTOR. The MCC is influenced by an ‘expert’ subcommittee referred to as the Complementary Medicines Committee (CMC). This committee was appointed by selective invitation and by no official public notice. This committee is made up of senior members of the Allied Health Professions Council and MCC officials. The AHPCSA has a vested interest in limiting the public’s over-the-counter access to natural health products, in favour of having them prescribed after the payment consultation fees. There are no members on this committee which represent therapeutic nutrition, manufacturing or integrative medicine.
- LIMITING OF PERSONAL FREEDOM OF CHOICE. The regulations severely limit our freedom of choice in healthcare by undermining section 12(2) of the Constitution. The State may not arbitrarily infringe on the rights of individuals to maintain their own bodily and psychological integrity, which includes the right to security and control over their own bodies.
- LIMITATION OF RELIGIOUS AND BELIEF RIGHTS. The regulations will block access to natural health products which may be integral to religious traditions, belief systems and cultural traditions. This is inconsistent with Section 15(1) of the Constitution.
- PURPOSEFULLY & WILLFULLY OBSTRUCTIVE. The regulations create impossible conditions for the majority of local small and medium sized companies to comply with. Despite warnings from the industry being relayed to the MCC about the consequences of inappropriate regulations and pharmaceutical standards imposed on their businesses, the MCC has ignored all these legitimate concerns.
- FATALLY CONCEIVED. There were no risk-benefit, cost-benefit or impact studies performed before the regulations were gazetted (or their subsequent amendments). The MCC lacks the capacity to evaluate the 110 000 products now it wishes to register. The administration must first prioritise the high-risk pharmaceutical drug registration backlog, before expending limited resources on low-risk natural health products.
- ENFORCING TRADE BARRIERS. The regulations severely impact on foreign natural health product imports and exports.
- NEGATIVELY IMPACTING ON EMPLOYMENT. The natural health product sector employs tens of thousands of people directly and indirectly. The regulatory regime will result in job losses for the majority of these people. Not only will the CAM companies be eradicated, but associated business sectors (raw ingredient suppliers, media advertising, packaging, logistics, printers etc). Approximately R9 billion of taxable income will be lost to the fiscus.
- NOT ENOUGH PHARMACISTS. In terms of the Medicines and Related Substances Act and the Pharmacy Act, all manufacturers or distributes natural health products will have to employ qualified and licenced pharmacists full time to oversee product manufacture and handling. Presently there is a shortage of at least 5 000 pharmacists in the State health sector with only 300 and 400 pharmacists graduating each year. A large percentage of these leave our shores on completion of their practical training year working for the State.
- UNDERMINES PUBLIC HEALTH. Natural health products and food supplements contribute to the health and wellbeing of the population, and in many cases prevent costly chronic and degenerative disease management. The setting of capricious daily limits on the ingredients of many of these products and regulating them out of efficacy (and potentially existence) will increase the country’s disease burden and the associated costs to the public health system in the medium and long term. Foreign research has proven that these products save billions of dollars of government spending due to their preventative and restorative functions.
A CALL TO ACTION!
Stakeholders can no longer be silent bystanders. They must determine their future and challenge these machinations of our drug regulator. The Medicines and Related Substances Act is a law of general application. The limitations, inequality and inappropriate regulation of natural health products by the MCC are unreasonable, unjustifiable and incompatible with a free an open and democratic society based on human dignity, equality and freedom.
There is an onus on the state to prove that all these limitations based on the regulation of high risk pharmaceutical drugs are indeed justified or in the public interest. In the words of Thomas Jefferson, “When injustice becomes law, resistance becomes duty”.
We invite you or your company to join the only organization in South Africa opposing and resisting these regulations, and which cares for yours and our future generation’s health freedom.
If you are a manufacturer, raw ingredient supplier, importer or wholesaler of natural health products, please (a) download our Prospectus, (b) fill in our online Manufacturing Sector Membership Form and (c), pay your annual membership fee.
If you are a retailer, direct seller or member of the public, fill in our General Membership Form and become a member today. If you wish to make a donation towards our work, make use of our secure PayPal donation facility on our website homepage at http://www.naturalhealthalliance.co.za.
If you represent a national or international association or organization with shared goals wishing to become an official Alliance Partner of the TNHA, feel free to contact us at contact@naturalhealthaliance.co.za.
