HERBAL HEALTH PRODUCTS: SHOULD THEY BE REGULATED AS FOODS, DRUGS OR AS A NEW CATEGORY?
HERBAL HEALTH PRODUCTS: SHOULD THEY BE REGULATED AS FOODS, DRUGS OR AS A NEW CATEGORY?
Anthony Rees
(General-Secretary: TNHA)
As a person who has practiced herbal medicine clinically, and worked in the natural health retail sector, and even owned a herbal health product companies over the last twenty years, I often pondered on whether herbal health products were merely unprocessed drugs, a segment of the food matrix, or whether they are unique substances which need to be defined and regulated separately. I have steadily come to the conclusion that for the most part, herbal health products fit somewhere between what we regard as foods and what we understand to be drugs. It is this vast grey area in-between which needs to be properly defined and appropriately regulated in South Africa.

THE SOUTH AFRICAN HERBAL MARKET
Millions of South Africans consume herbal health products, whether as single ingredients or as multi-ingredient formulas.
They are made up of either African traditional herbal ingredients (indigenous plants) or non-indigenous herbal ingredients derived from herbal traditions abroad.
Research conducted at African traditional herbal markets around the country, such as the African medicinal plant market alongside Durban’s Grey St and Warwick Avenue reveals that there is no slowing down in the consumption of African traditional herbal products by black South Africans, despite the expanded footprint of Western primary healthcare after apartheid.
It is estimated that between 70% of black South Africans first utilise African traditional medicine before seeking Western medical treatment.
In the more formal health store and pharmacy market there has been a surge in the sales of non-indigenous herbal products over the past two decades, mirroring European, Australian and North American herbal industry trends.
Many people are seeking natural health products where they believe “conventional” medicine has failed due to unwanted side effects, lack of efficacy and the high cost of drugs. Herbal health products now account for a multi-billion Rand market.
HERBALISM IS OF A DIFFERENT PARADIGM TO ORTODOX MEDICINE
The regulation of herbal products and has been caught up in a bitter battle of paradigms, caught between those who believe that they should be regulated as drugs and those who believe that they should be regulated as foods. This debate has dragged on for several decades without any consensus being reached globally.
I believe that the words “medical” or “medicine” have been usurped by the disease management sector in our modern era, and refer to pharmaceutical drugs or remedies administered to alleviate symptoms.
Nutrition deals with foods and food-derived isolates, taking in nourishment so that the body grows, produces energy, repairs, heals, and maintains itself.
The vast majority of herbal remedies are caught in a large grey area between these two systems, with some overlapping on either side.

A growing body of research indicates that many plants have “therapeutic and preventative potential” in their natural, unprocessed state. Some can present unwanted side effects and some can be extremely toxic, however the vast majority are of low risk and safe to consume.
Drug regulatory authorities around the world have defined herbal health products as either dietary supplements (foods) or as medicines (drugs).
Some regulators, like the US Food and Drug Administration (FDA) have classified all herbal health products as ‘dietary supplements’, fitting squarely in the food category, while others like the German Federal Institute for Consumer Health Protection and Veterinary Medicine and the Danish Health and Medicines Agency have classified them as pharmaceutical drug, requiring similar assessment and licencing requirements to drugs. The former have a thriving innovative market, and the latter allow only a few approved herbal products, and often only with a doctor’s prescription.
This ambiguity has resulted in a tug-of-war in countries attempting to draft regulations for herbal remedies, where the industry prefers a food-based regulatory regime, due to the low risk nature of the products and relaxed compliance requirements, and the pharmaceutical regulators who view them as ‘crude drugs’ requiring restricted access.
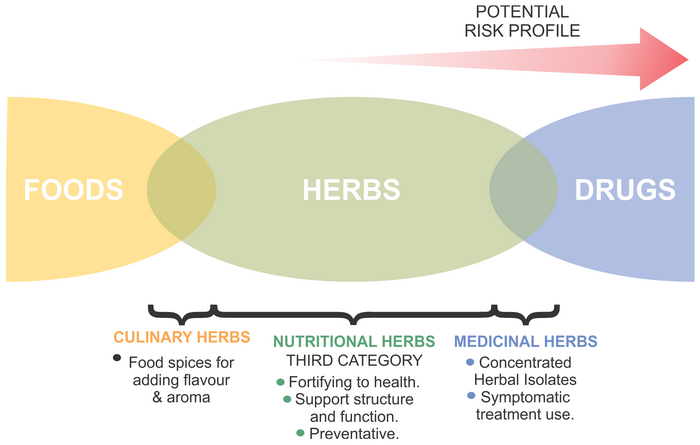
Some countries like Australia, Canada and Singapore have adopted a ‘THIRD CATEGORY’ compromise, by clearly defining and appropriately regulating the grey area between foods and drugs in an innovative way. Despite the South African Medicines Control Council having created a separate ‘Category D’ in regulations published in November 2013 for natural health products, the licencing requirements for manufacturing facilities and application process to register natural health products is no different to what is required to manufacture, licence and distribute pharmaceutical drugs.
HERBS ARE DIVIDED INTO THREE MAIN CATEGORIES
It has become clear to me over time that herbal products can be broken down into three basic categories.
- CULINARY HERBS: The food use of herbs as vegetable products for adding flavour or aroma to food.
- NUTRITIONAL HERBS: The botanical view that defines herbs as non-woody, seed-producing plants which provide plant nutrients, are fortifying to health, support structure and function in the body and prevent dis-ease.
- MEDICINAL HERBS: Herbs that contain symptom treating actions, or highly concentrated isolated chemicals from plants which are drug-like in their actions.
These three categories can interrelate depending on the way herbs are processed, how they are consumed or the amount (dosage) used. Herbs in the first two examples are nutritional. Some argue that the third example, medicinal herbs, should also be considered nutritional because they are “natural” and don’t represent synthetic, novel drugs at all.
Modern society demands a quick fix and wants whatever has been ingested or rubbed on the skin to have an immediate effect, whether it is relief from pain, the ability to sleep, have a bowel movement, feel energy, be released from depression etc. This is much easier than taking the responsibility, time and effort to make lifestyle changes, dietary improvements and other adjustments that bring about real sustained health and wellness.
Some confusion arises because the difference between a food-herb and a medicinal herb is not always clear cut. For example, sulphur compounds in garlic (Allium sativum) support the body’s immune response, the inflammation and repair process. This is a nutritional effect; the biochemistry is not stimulated or suppressed; there is no masking of symptoms. However, if the sulphur were isolated and highly concentrated, then it would have more of a pharmacological – stimulating or suppressing or masking action, leaning towards a drug.
Another example is the salicylates found in tiny amounts in tomatoes (Solanum lycopersicum), apples (Malus domestica), cucumbers (Cucumis sativus), White Willow bark (Salix alba), Wintergreen (Gaultheria procumbens), and other foods and herbs. These appear in complexes with a multitude of other co-factors and nutrients. But once isolated or synthesized in a laboratory, salicylates become Aspirin, a drug. Most pain relievers contain 300 milligrams (mg) or more of salicylates.
Most food sources contain between 1 to 4 mg per 100 grams; an apple contains about 2 mg. There is a huge difference between the apple and two aspirin tablets (600 mg salicylates).
How many apples could one eat to ease a headache? Not only would it not have the pharmacological effect because it is a food, but it would be more than uncomfortable to eat the number of apples needed to get 600 mg of salicylates (300 apples)! Nature incorporates built-in safety modes.
Nutrients can prevent or reverse the course of dis-eases: Vitamin C (ascorbic acid) for scurvy, Vitamin B-1 (thiamine) for beriberi, Vitamin B-3 (niacinamide) for pellagra, Vitamin D complex for rickets, etc.
But this should not be confused with the pharmacological action of penicillin for an “infection” – interference in the biochemistry of inflammation and repair – or other drug reactions.
“Nutrients aid and enhance the body’s own biochemical efforts and natural processes. Drugs interfere with biochemistry, provoking or restraining normal responses”HERBS AS DRUGS
Many modern drugs had their origin as herbs, plants, or other natural substances. These materials are vigorously analysed, tested, and then condensed to an extract of the so-called “active ingredient(s)”.
The pharmaceutical industry has isolated the “active ingredient”, like Aspirin from the inner bark of the White Willow tree (Salix alba) or codeine from the Opium Poppy (Papaver somniferum) and synthesized it (artificially imitated), often bypassing any need to use the actual plant.
In order for pharmaceutical companies to profit from these substances they first need to patent them. This requires that they need to chemically alter the naturally occurring active ingredients so that (a) they continue to work in the same or similar way, and (b) can be inexpensively reproduced by chemical synthesis or more modern biotechnological means.
Aspirin is an example of how the pharmaceutical industry turns a naturally occurring molecule found in plants into a patent drug, the world’s most common drug. In the below graphic (left) you can see the original salicylic-acid molecule derived from the bark of the White Willow tree (Salix alba) and the Meadowsweet herb (Filipendula ulmaria). On the right is the pharmaceutical version of salicylic acid, now termed acetylsalicylic-acid, first patented by Bayer in 1939. Salicylic acid is now biosynthesized and no longer has any plant base.

Foxglove (Digitalis purpurea) contains cardiac glycosides (especially digitoxin and digoxin) which were used to treat heart disease by increasing contractility of the cardiac muscle and thus cardiac output. The plant molecules are no longer utilised in their natural form.
Like Aspirin, the pharmaceutical industry patented new molecules based on dioxin and dixitoxin. Two drugs now sold in the pharmaceutical market have been produced synthetically which differ in that digoxin has an additional hydroxyl group at the C-3 position on the B-ring (adjacent to the pentane). Both molecules now include a lactone and a triple-repeating sugar called a glycoside.
“Digitalis,” used to designate the whole group of glycosides, is, like all drugs, a poison and can have adverse effects.
Most toxic plants, or those which are dangerous at higher dosages have been identified over the years, and regulators around the world have either banned their use outright, or restricted their use by scheduling them so that only trained professionals can prescribe them. For the most part, herbs commonly sold by natural health product manufacturers today are officially classified as ‘Generally Regarded As Safe’ (GRAS).
MOST HERBS SOLD IN COMMERCIAL NATURAL HEALTH PRODUCTS ARE NOT DRUG-LIKE
Nature created plants with a plethora of interacting constituents that not only produce actions or reactions, but which also balance, protect, and nourish the body on the cellular level. From healthy cells come healthy tissues, healthy organs and healthy body systems.
The “active ingredients” of plants are not concentrated and never appear alone in whole-herb form, so they have “less potential to cause side effects or serious overdoses.” White willow bark, for instance, contains salicylates which are not isolated or concentrated enough to cause gastrointestinal bleeding (as will the Aspirin drug), and one’s stomach will not hold enough of it to cause an overdose.
Hippocrates, the father of modern medicine and Galen, the first chemist knew White Willow bark could ease aches and pains and reduce fevers. It has long been used in Europe, the Middle East and China for pain relief. The Cherokee and other Native Americans used an infusion of the White Willow bark for fever and other medicinal purposes too.
Yet with modern technologies, the salicylates can be isolated and concentrated, then combined with some of the original herb and then called “natural”. Though not designated as a “drug,” a line surely must be drawn between what is nutritional and pharmaceutical.
HERBAL SIDE EFFECTS AND CONTRAINDICATIONS
Although “medicinal” herbs are gentler than novel synthetic drugs, some may cause side effects and are not always safe in any amount. Any substance, whether natural or not, that is strong enough to act as a medicine can be taken inappropriately and can cause unwanted side effects.
‘Sola Dosis Facit Venenum’ is Latin for ‘The dose makes the poison’. This adage was coined by Paracelsus, philosopher, physician and botanist in the early sixteenth century. The principle relies on the finding that all chemicals — even water and oxygen — can be toxic if too much is eaten, drunk, or absorbed through the skin. Herbs are not excluded from this rule.
Goldenseal (Hydrastis canidensis), for example, taken for long periods of time or in large amounts (depending on individual tolerance) can cause damage to the liver. Many herbs are safe for regular use, yet may pose some risks during pregnancy. Examples include Buckthorn (Cascara sagrada), Coltsfoot (Tussilago farfara), feverfew (Tanacetum parthenium), Senna (Cassia senna), and certain concentrated essential oils like Pennyroyal (Mentha pulegium) etc.
In most cases, if a side-effect or contraindication is experienced, removing the herb from the treatment regimen or food will reverse the disorder quickly and without any long term damage. These effects are reported less frequently nowadays, as label warnings are placed on products by responsible manufactures and because information technology has created a savvier consumer. A good list of herb contraindication can be found here: http://tinyurl.com/zhjss34
HERB-DRUG INTERACTIONS
We all know that before we take new pharmaceutical drug, we should first make sure our medical doctor and pharmacist knows about all the other medications we’re taking to avoid potential interactions that can change how our medications work.
What most people don’t know is that it is equally important that we consider the potential interactions between our medications and herbal remedies. Some common remedies have potent effects that can interfere with both prescription and non-prescription medications. Some herbal supplements may decrease a pharmaceutical drug’s effectiveness or, conversely, amplify the effects to a dangerous level. These may also increase the risk of experiencing drug side effects. If a doctor is not interested in herbal remedies (probably due to his/her lack of knowledge on their usage) or is not willing to address questions about them, you can contact a registered Phytotherapist (Medical Herbalist) who is professionally trained in the safe use of herbs through the South African Association Of Herbal Practitioners (http://herbalpractitionerssa.co.za). You can also study this informative herb-drug interaction chart (http://tinyurl.com/zxupwcw)
“CAVEAT EMPTOR!” (BUYER BEWARE!)
I strongly believe it is not always necessary for drug regulators to ban or restrict herbs which may pose potential side-effects, contraindications or herb-drug interactions, if appropriate labelling warnings can be made mandatory as in the case of most over-the-counter (OTC) pharmaceutical drugs, cigarettes and even alcohol for that matter.
Appropriate label warnings can direct vulnerable groups or individuals (e.g. pregnant women) to refrain from using herbs identified which may cause harm, or limit the daily dosage to what is regarded as a ‘safe dosage level’.
To summarize, some herbs are more drug-like (not foods) to be used as treatment for the short duration of illness or injury. These are usually prescribed symptomatically. Other herbs are more food-like that can be used daily, but in smaller amounts.
For instance, garlic (Allium sativum) and parsley (Petroselinum crispum) are food herbs and are consumed regularly by many people throughout their lives. Yet one could not eat 500 grams of garlic or a kilogram of parsley – the intensity of the taste or aroma, reflecting the intensity of the food components, prevents it. This is Nature’s way of saying these herbs should be used in tiny amounts and to enhance other foods and their nutritive value.
THE EXACT WORKINGS OF MOST HERBAL REMEDIES REMAINS A MYSTERY SO SCIENCE AND REGULATORS
Plants manufacture complex and numerous diverse compounds which work together to make “a remedy that is more than the sum of its active ingredients.” This phenomenon is called synergy. Some of the supportive compounds may be important to reduce the possible toxicity of active constituents, or act as preservatives, or enhance the efficacy upon the body, among its other roles.
Like food, once those ingredients are separated or synthesized, the comprehensive function – the biological activity – is lost and the body can use it only as a drug, a xenobiotic (foreign substance).
Drugs focus “on blocking a single pathological biochemical / molecular pathway,” while whole herbs affect a variety of pathways and are naturally designed to cope with biochemical complexity.
Herbs hold the potential for greater clinical gains (results) than drugs because of the likelihood of additive and synergistic effects. This permits low dosage levels, which, combined with the already modest concentrations of most phyto-chemicals (plant active ingredients), greatly minimizes the risk of side effects.
In contrast, the pharmacological application of herbs employs a dose that will induce strong drug-like effects. The aim is quick, temporary relief for an acute condition such as insomnia, a cold, or pain. The herb is used “to override the physical condition”. The goal is directed at the effect, not the cause. In many ways, herbs used in this way are being used no differently to all pharmaceutical drugs (symptomatic treatment).
Side effects (unwanted effects) and toxic reactions are more of a risk, especially due to large doses, extended duration of use, or individual sensitivities. Symptoms are masked or overridden, but true healing may not occur. Pharmacological doses of herbs are best used for relief of acute conditions rather than chronic ones, and should only be used on a short-term basis, for a few days or up to a week.
Physiological (tonic) use of herbs applies to herbs and doses that support, rather than override, normal biochemical function and support the body’s own self-healing mechanisms. Both the individual’s and the herb’s uniqueness are considered so the minimum amount required to produce an effect is administered.
This type of herbal use can be applied to most chronic conditions and diseases of lifestyle, since it is safer and more nutritionally-oriented to use them for longer periods of time. Even herbs with potentially strong effects can often be taken with this method. The “downside” to this approach is that it must be combined with other natural healing modes including dietary adjustments, the possible use of food supplements, rest, or exercise. It means time and effort, not the ease of taking “a pill for every ill” to make a symptom abate.
Because they act gently, most herbal products sold in health shops and pharmacies will not have dramatic or immediate effects. If they do, then they are medicinal herbs or highly concentrated (usually containing isolated active ingredients), with the corresponding risk of side-effects or biochemical imbalances.
Some herbs gradually improve health by encouraging organ or endocrine gland function, digestion, elimination, detoxification, etc, so the body is better able to repair or heal. Herbs can nourish the immune system; aid regeneration of damaged liver tissue; invigorate heart muscle; help build up the adrenal, thyroid, reproductive, or other glands; improve breast milk production; soothe the gastrointestinal tract; relax blood vessels so they dilate properly and support musculoskeletal tissues. Health claims made for these uses are called ‘structure-function claims’, and are allowed in various progressive countries.
A NOTE ON ‘STANDARDISED’ HERBAL PRODUCTS
“The idea that the medicinal effect of an herb is due mainly to a single active ingredient is a concept that is well ingrained in modern pharmacology,” says Alan R. Gaby, MD.
This pharmacological viewpoint is reflected in the trend toward standardization of herbs which is based on the concentration of a specific, isolated ingredient.
To “cope” with the normal inconsistency of “active ingredients” in herbs (from growing in different soils, being picked in different seasons, varying harvesting procedures, being subject to different processing methods etc.), standardization places a minimum concentration of these particular constituents in a product. That is, the manufacturer “guarantees” that the herb contains a specific amount of a certain chemical (active ingredient).
“Guaranteed potency” herbs are standardized to insure a specified amount and the activity of the desired constituent(s). Isolated extracts of the so-called “active ingredient(s)” are often added to the original herb and in many cases a synthetic chemical is used.
Standardized herbs have been heavily promoted as being more potent and containing a more reliable dose than their whole herb counterparts. This discounts the complex, balanced, synergistic mixture of ingredients which act together for physiological and nutritional benefits as well as for reduced toxicity. For example, an overdose of the drug digitalis causes severe cardiac arrhythmia. But when the plant source – foxglove – is taken to excess, nausea occurs as an early warning sign long before any cardiac arrhythmia can develop. Natural compounds in the foxglove reduce likelihood of toxicity. But the single “active ingredient” – digitalis – is always much more toxic.
The “active ingredient” does not always produce the expected action.
The hypericin active ingredient in St. John’s Wort (hypericum perforatum) had long been thought to account for its anti-depressant effect, so its products are often standardized for hypericin content. More recent studies have demonstrated that the beneficial effect does not depend on the content of hypericin, as the remainder of the herb was as efficacious as the active ingredient earlier identified. Therefore, the sum of the parts does not necessarily equal the function of the whole.
An extraction method that maximizes the content of one compound can and does remove other beneficial substances. The whole cooperative complex in whole herbs can produce nutritional benefits. While “a modern reductionist approach to herbal remedies” may be desirable for a pharmaceutical effect, it should not be confused with a tonic or nutritional effect.
Pharmaceuticals are strong, isolated chemicals with profound effects for masking symptoms. The herbal concept is supposedly alternative to this drug method. Why should herbs be made into drugs and then administered as “natural” health products? There is only one answer ……. Because they easier to sell (for a quicker fix), and at a huge profit!
HERBAL SAFETY – WHERE ARE ALL THE DEAD BODIES?
One of chief motivations used by drug regulators across the globe to bring herbal remedies and other natural health substances under full pharmaceutical regulation has been the question related to ‘SAFETY’.
There have been many calls, especially from prominent individuals aligned with the pharmaceutical industry or who have previously served as drug regulators, to clamp-down on the sale of herbal products, citing a few isolated reports of adverse reactions, contraindications or even deaths related to their use. They have also propped up special interest groups who manipulate the media to embark on campaigns to vilify natural health products at every opportunity.
These reports, when scrutinised carefully, prove to be nothing more than ‘red herrings’ to steer public opinion against non-drug therapies and undermine their confidence in natural health products. They are also designed to motivate regulators and legislatures to usher in restrictive laws to severely curtail and even destroy the growing natural health industry.
Adverse reactions to prescription and over-the counter drugs kill at least people 200 000 people in the USA each year and are officially the third leading cause of death in the developed world after heart disease and cancer. Non-fatal poisonings in the USA run at about 500 000 reported incidents ach year.
The World Health Organization has managed to come up with only 5 000 cases of “suspected” adverse reactions to commercially sold herbs, yet the time period and geographical range has never been revealed. Most of the adverse reactions and even a handful of deaths attributed to commercial herbal products are due to misuse because they are either mislabelled, contaminated with pharmaceutical drugs (mainly through illicit imports from the Far-East) or come with no safety information.
According to statistics recorded at the Red Cross War Memorial Children’s Hospital in Cape Town, between 2003 and 2008, there were only 43 admissions to the hospital’s casualty department for adverse reactions after children ingested herbal and nutritional supplements (multi vitamins). All survived and were soon discharged after being admitted. The Red Cross Africa’s War Memorial Children’s Hospital is largest referral hospital for children on the African sub-continent.
The South African National Adverse Drug Monitoring Centre, run the University of Cape Town for the Medicines Control Council has no historical data related to adverse reactions or deaths related to commercial herbal remedies or other nutritional supplements despite them recording Adverse Drug Reactions (ADRs) for over three decades.
The American Association of Poison Control Centres has reported ADRs in the Journal of Clinical Toxicology each year for decades. In recent years there were no deaths reported which related to severe adverse reactions resulting from herbal remedies and other natural health products. This is despite an estimated six billion single doses of natural health products being consumed by American consumers each year.
Most commercial herbal remedies sold in health shops nowadays are extremely safe. They are even statistically safer than commercial foods we buy the supermarket.SAFETY AND THE BURDEN OF PROOF FOR HARM
According to the US Dietary Supplement Health and Education Act of 1994, herbal remedies cannot be removed from the marketplace until they have been proven to be harmful to the public. The burden of proof to demonstrate harm is placed squarely on the Food and Drug Administration and other consumer protection agencies.
In other countries, herbal products cannot be sold to the public unless the manufacturers, importers and distributors can first prove that they are safe. This type of regulatory system for natural health products creates a costly and unnecessary burden on industry and the regulators. The vigorous and stringent pharmaceutical testing required to prove the safety of herbal remedies prohibits small and medium sized companies from selling their intended products, and/or results in consumers having to carry the costs, making the products unaffordable. Sadly our South African drug regulator, the Medicines Control Council, has embarked on a regulatory regime that places the burden of proof for safety of herbal products in the hands producers.
KEEPING THINGS SIMPLE
“Herbal medicine is like any other medicine,” says James Duke, Ph.D. “It should be used with discretion.”
One way to view the relative safety of herbs, he says, is to compare them to coffee. He rates herbal teas according to whether he views them more or less safe than coffee, which he would limit to two cups a day (more can cause minor to major side effects). Water hemlock (Cicuta maculata & C . bulbifera) and Belladonna, known to be deadly, would not be touched. Goldenseal (Hydrastis canadensis) is to be limited to one cup a day. He considers Lemon Verbena (Aloysia citrodora) as he would coffee and may drink two cups a day. He would be afraid to drink three cups of Feverfew (Tanacetum parthenium) or Chamomile (Matricaria chamomilla).
AN APPROPRIATE ‘THIRD CATEGORY’ REGULATORY SYSTEM IS REQUIRED IN SOUTH AFRICA
It is safe to say that any potential regulatory system put into place to regulate herbal remedies will be faced with challenges in balancing the consumer’s right to access the health interventions of their choice, and protecting them from possible harm.
Perhaps the simplest way to regulate these low risk products (the vast majority of natural health products), would be to create a third category, and not treat them exclusively as either foods or drugs. For the most part, herbal remedies sold in today’s health shops and pharmacies are neither drug-like nor food-like. They fall somewhere in-between.
The TNHA will soon embark on drafting a draft Traditional & Natural Health Products legislative proposal, which will clearly define traditional and natural health products separate from “medicines” (drugs) under the Medicines & Related Substances Control Act (Act 101 of 1965), and foods under the Foods, Cosmetics and Disinfectants Act (Act 54 of 1962). It will also propose an appropriate regulatory system and new statutory council for to regulate them accordingly.
